A color change can represent a chemical change (asĮxplained later).  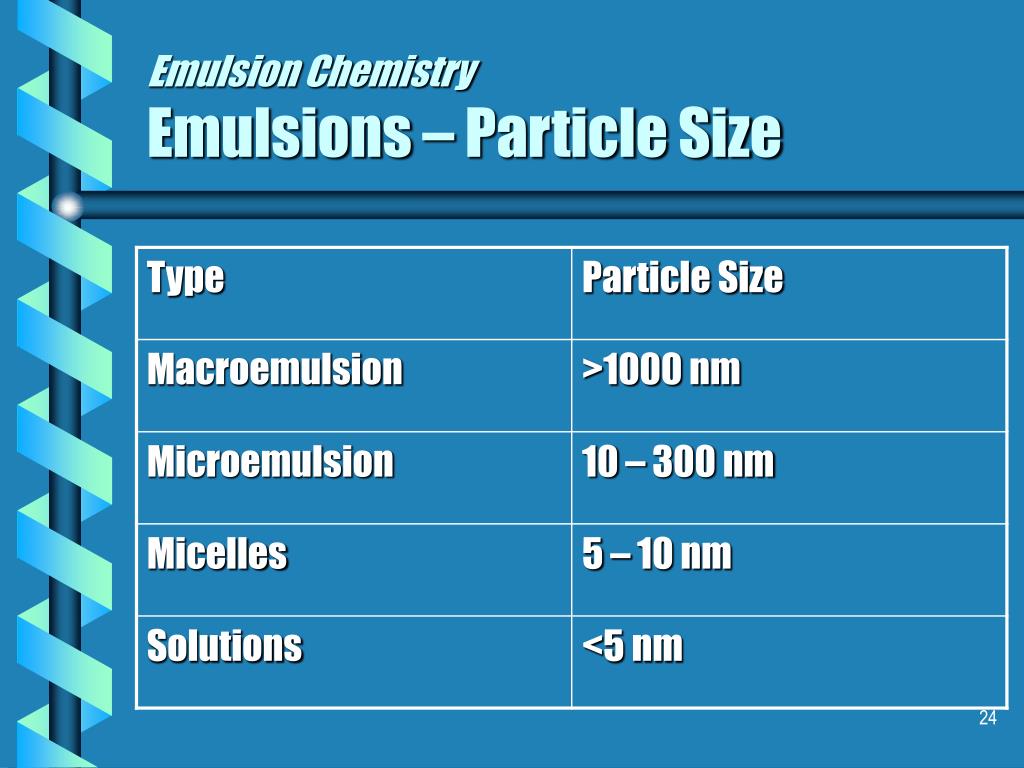
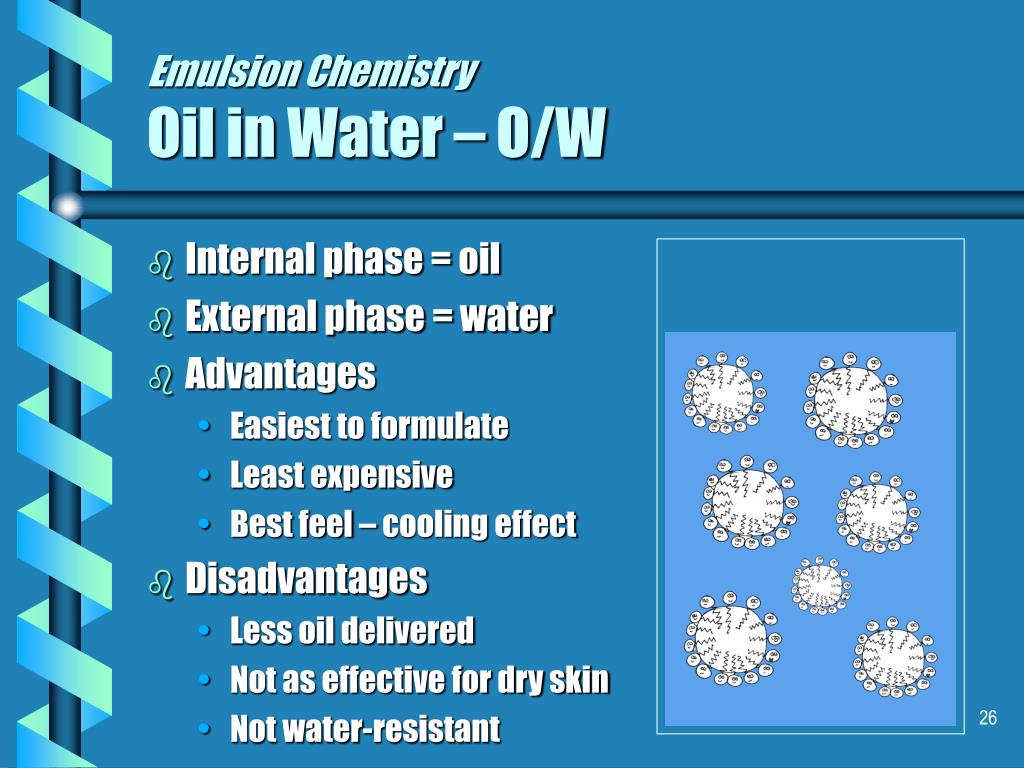
In lesson #4 turns white as the oil and water are mixed in the presence (the darker line in the illustration below). A light beam passing throughĪ solution can be seen only along the direction of original incidence Homogenous mixtures) do not scatter light. That most clouds and fog appear white is also the result of the Tyndall Scatters some of the light from a movie or slide projector. Many of us have seen this already as the dust in the room (sometimes it could take years to separate the oil and water in anĮffect is defined as the scattering of light by the particles of a Lengthen the amount of time it takes to separate these two phases They separate almost immediately afterward.Īlthough you can create an emulsion without a surfactant, surfactants Italian dressing knows, the oil and water do not mix unless shaken Temporarily stabilize the oil and water mixture. Surfactants lower the surface tension of the water, thus making itĮasier for cleaners to lift dirt and grease off of dirty dishes andĬlothes and help to keep them suspended in the dirty water to keep themįrom being redeposited on the clothes and dishes. Soap, but other examples of surfactants include detergents, hand soaps,Įtc. The surfactant we used in all of the following experiments is dish It is said that they "make water wetter". A surfactant can be consideredĪ "confused" molecule having both a hydrophobic (water-hating) and In an oil continuos emulsion, oil is theīulk of the material and the water is the dispersed phase (see diagramsĪre used in the emulsification process. InĪ water-continuous emulsion, the water is the bulk of the material and Kinds of emulsions: water continuous (o/w) and oil continuous (w/o). Sunscreens, cosmetics, salad dressings, condiments, hand creams, etc. They are the basis of countless household products such as: In nature but few people really understand exactly what emulsions are.Įmulsions are classified as a colloid (see concept map below).Įmulsions are at least one fluid (liquid) dispersed in another fluid Accurately record data using metric system units.Evaluate the emulsion type of various commercial products.Analyze commercial products to determine emulsion type.Differentiate between a water-continuous and oil-continuous emulsion.Test for a chemical change by using pH paper.Explain the role of surfactants in the emulsion making-process.Synthesize a water in oil "w/o" emulsion.Determine the relationship between label ingredient order and emulsion type.Use the scientific method to determine which common household detergent creates the most stable emulsion.Use the scientific method to determine the best ratio of oil to water in making an emulsion.Realize that emulsions are the basis of many common household products.Apply the skills of observation, data collection, and scientific reasoning.Understand the Tyndall Effect in emulsions.Compare and contrast chemical and physical changes.Observe the behavior of the interaction of liquids of different densities.Create an emulsion from common household products.Understand what an emulsion is and how it fits into the classification of matter.Upon completion of this lesson the student will be able to: Using emulsions to teach the scientific method, matter classification, and chemical/physical changes.įormulation Chemistry, Rohm & Haas Company Emulsion K-12 Experiments & Background Information For Science Labs, Lesson Plans, Class Activities & Science Fair Projects For Middle School Students & Teachers
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
